|
This part of the spectrum is called the fingerprint region. The table lists IR spectroscopy frequency ranges. 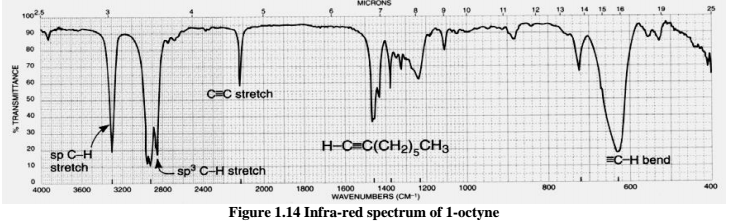
You will notice that there are many additional peaks in this spectrum in the longer-wavelength 400 -1400 cm -1 region. The IR Spectrum Table is a chart for use during infrared spectroscopy. Similarly, bonds to lighter atoms (in particular to hydrogen) vibrate at higher frequency than bonds to heavier atoms. Shorter and stronger bonds have stretching vibrations at higher frequencies than longer and weaker bonds. Nevertheless, it can serve as a familiar reference point to orient yourself in a spectrum. symmetrically substituted alkene CC double bond) may not absorb at all. This peak is not terribly useful, as just about every organic molecule that you will have occasion to analyze has these bonds. The jagged peak at approximately 2900-3000 cm -1 is characteristic of tetrahedral carbon-hydrogen bonds. Within that range, carboxylic acids, esters, ketones, and aldehydes tend to absorb in the shorter wavelength end (1700-1750 cm-1), while conjugated unsaturated ketones and amides tend to absorb on the longer wavelength end (1650-1700 cm -1). Notice how strong this peak is, relative to the others on the spectrum: a strong peak in the 1650-1750 cm -1 region is a dead giveaway for the presence of a carbonyl group. The key absorption peak in this spectrum is that from the carbonyl double bond, at 1716 cm -1 (corresponding to a wavelength of 5.86 mm, a frequency of 5.15 x 10 13 Hz, and a ΔE value of 4.91 kcal/mol). However, the utility of the fingerprint region is that the many bands there provide a fingerprint for a molecule. The fingerprint region is often the most complex and confusing region to interpret, and is usually the last section of a spectrum to be interpreted. conjugated alkene CC stretching 1650-1600 medium conjugated anhydride CO stretching 1775 strong. Many different vibrations, including C-O, C-C and C-N single bond stretches, C-H bending vibrations, and some bands due to benzene rings are found in this region. This region is notable for the large number of infrared bands that are found there. How To Interpret IR Spectra In 1 Minute Or Less: The 2 Most Important Things To Look For Tongue and Sword Last post, we briefly introduced the concept of bond vibrations, and we saw that we can think of covalent bonds as a bit like balls and springs: the springs vibrate, and each one sings at a characteristic frequency, which depends on the strength of the bond and on the masses of the. The region of the infrared spectrum from 1200 to 700 cm -1 is called the fingerprint region. Group frequency and fingerprint regions of the mid-infrared spectrum These types of infrared bands are called group frequencies because they tell us about the presence or absence of specific functional groups in a sample. This makes these bands diagnostic markers for the presence of a functional group in a sample. For example, C-H stretching vibrations usually appear between 32cm -1 and carbonyl(C=O) stretching vibrations usually appear between 18cm -1. \)Īn important observation made by early researchers is that many functional group absorb infrared radiation at about the same wavenumber, regardless of the structure of the rest of the molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
